Sodium is a chemical element with the symbol Na (from Latin: natrium) in the periodic table and atomic number 11. It is a soft, silvery-white, highly reactive metal and is a member of the alkali metals; its only stable isotope is 23Na. The free metal does not occur in nature, but instead must be prepared from its compounds; it was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide.
Given its extraordinary chemical reactivity, there are multiple applications for this substance. Preparing it via electrolysis is the easiest way to go, however we need to use a molten sodium hydroxide electrolyte, as the resulting sodium would react with the water in an aqueous solution. Not that hard to do, as NaOH melts at 318 °C . The temperature is reasonably low to allow the separation of the resulting sodium without spontaneous ignition in air, but extra caution must be exercised as the chemicals involved are highly caustic, especially at high temperatures.
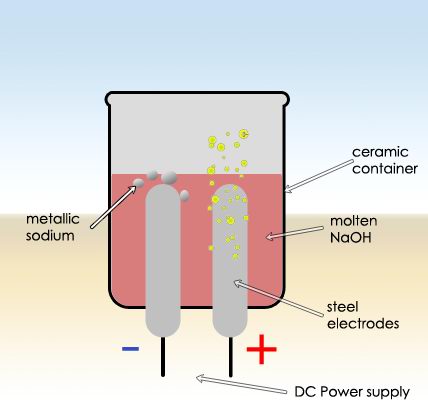
I used a ceramic electrolysis container with two steel electrodes. Carbon rods doesn’t go to well. The power supply was my trusty variable regulated DC supply, and most of the time I used 7-10V. To melt the sodium hydroxide I used a butane torch: melting is necessary to get the electrolysis going , but then the flowing current will keep the NaOH in molten state and the torch is not required anymore.
In the laboratory, with carefully controlled conditions, sodium metal can be isolated from the electrolysis of the molten monohydrate in a low temperature version of the Castner process, according to the following reaction:
4 NaOH·H2O(l) → 4 Na(l) + O2(g) + 6 H2O(g)
The monohydrate does not need to be heated in order to melt, as the process produces enough heat due to ohmic heating. However, it must be initiated with a small quantity of liquid water to create an electrically conductive electrolyte. As the system’s temperature increases, the monohydrate will start to melt at about 65 °C as stated above. Only when the temperature reaches about 100 °C can sodium be isolated. Below this temperature, the water produced will react with the sodium: above this point, any water formed will be driven off in the vapour phase, creating an essentially anhydrous reaction. While this process has some advantages over other electrolytic processes, it is not preferred by most chemists because a marginal quantity of sodium produced boils at the electrode interface.
If temperature is too high, part of the sodium will dissolve in the molten NaOH, as shown in the video (the black substance surrounding the metallic sodium spheres). Also the electrodes should be some space apart to prevent short-circuit due to the liquid sodium getting mixed in the electrolyte.
NaCl might be a good alternative, but at the high temperature needed to melt it, handling sodium is more problematic.
Eye protection is an absolute must, as the chemicals involved will quickly burn through skin!












I’m curious if potassium chloride might be used in place of the sodium chloride in the video. Potassium metal, when it interacts with water, creates a much more violent chemical reaction than does sodium. I presume the cathode would be where the potassium would collect as well.
@Ryan your assumptions are correct.
Si cum scoti Na fara sa arda?
Salut Dani 🙂 . Il las sa se raceasca.
Hi, how did you fill the space between electrodes and the ceramic container? I can’t get an idea which material is suitable for such high temperature and aggressiveness of melted NaOH.